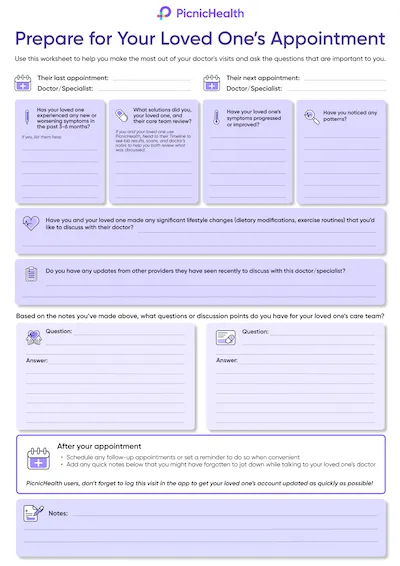
Last month, PicnicHealth welcomed Rachael Thomas Higgins to the team as our new Chief Commercial Officer. Rachael brings over 20 years of commercial expertise to PicnicHealth, most recently serving as Vice President of Commercial Sales at CorEvitas through its acquisition by Thermo Fisher Scientific. In her past roles, which also include leadership positions at CircuitClinical, TriNetX, and Covance, she has successfully defined go-to-market strategy and driven growth for life science solutions.
We recently sat down with Rachel to talk about her journey to PicnicHealth and her thoughts on the observational research space.
1. What sparked your interest in joining PicnicHealth?
I love what PicnicHealth is doing. The way observational research is done today is broken – not only making these types of studies complex and costly for life science companies, but also cumbersome to patients. Noga and Troy [PicnicHealth co-founders, and CEO and CTO, respectively] recognized that breaking down data silos can enable more streamlined and comprehensive research as well as give patients greater ownership of their health. Leveraging innovative technologies, the PicnicHealth team has been able to bring observational research into the future while keeping the patient at the forefront.
2. How do you think PicnicHealth is disrupting the observational research space?
Traditional methods are not always the best methods.This is especially true for observational research, which to date has often been run like a site-based intervention trial or with off-the-shelf data. These methods lead to high loss-to-follow up rates, data gaps, and burden on everyone involved.
PicnicHealth is uniquely able to virtually enroll a patient into a study and capture their full past and future health journey – from all of their clinical encounters across the United States. Since we consent patients into studies, we’re able to work directly with them to administer surveys as well as collect other forms of active data. Our data collection and curation methods also offer us the agility to adapt to evolving studies and explore new concepts effortlessly. Achieving this level of completeness and flexibility requires a highly sophisticated and purpose-built engine.
3. What does “innovation” mean to you and why is it important in observational research?
Innovation is the key to evolving. Observational research needs innovation so that it can be faster, more efficient, and less complex. This is why technology and artificial intelligence is a big focus for us at PicnicHealth; it enables us to do things like enroll patients from anywhere, gather insights from unstructured data, rapidly pull data from multiple sources, and monitor study progress. With these tools, our epidemiologists and data scientists are able to run observational studies more efficiently than was ever possible before.
4. Based on your experiences, how do you see the life science industry evolving over the next five years?
I see the role of technology continuing to increase. There is a growing need from stakeholders for more, high-quality evidence, and the go-to sources of data don’t have the granularity, longitudinality, or completeness to cut it anymore. At the same time, site-based study models are too expensive, time-consuming, and inflexible. Technology will be the key to improving the speed and accuracy of research.
Patient centricity will also continue to be an important factor in running studies. There are greater demands for representation in research to ensure that results are generalizable to the population that is affected. It’s also clear that patient questionnaires on quality of life measures add critical information to the understanding of diseases and treatments. Increasing diversity and engagement are only possible if research takes a patient-focused approach.
Want to hear more about how PicnicHealth is radically simplifying observational research? Meet Rachael at ISPOR on May 5-8, or set up some time to chat here.